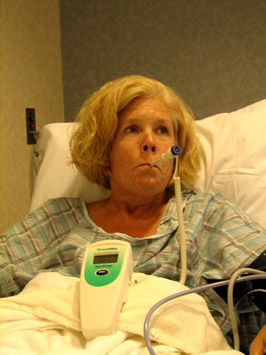
Last summer I began experiencing serious pain and tenderness on the right side of my body below my belly button. The advice nurse sent me directly to the emergency room suspecting appendicitis. While I was in the emergency room waiting on various tests, a patch of skin over the painful area began to turn red and increase in size.
“The problem is not your appendix,” said the doctor, “but a skin infection, and one that has to be dealt with right away.” So I was promptly hooked up to an antibiotic solution dripping from a bag hung on a nearby metal stand.
While watching the antibiotic drip, I asked the doctors whether my infection could be caused by Methicillin-resistant Stapholoccus aureus (MRSA), a notorious bacteria resistant to penicillin and other drugs. I was aware of MRSA because I work on antibiotic-resistant bacteria here at UCS.
Although MRSA had once been mostly a problem for ill people in hospitals, it is now showing up in healthy people like me. I also knew that these so-called community-acquired MRSA infections can be wicked. Newspapers over the last decade have been filled with stories of MRSA-skin infections killing healthy young people, including athletes.
Taking No Chances
My doctors weren’t sure my infection was caused by MRSA but nowadays they don’t take chances. “If a skin infection looks like MRSA, we treat it like MRSA.” They have every reason to be cautious. MRSA is estimated to cause 19,000 deaths a year, more than AIDS.
Where did this infection come from? There were no scratches or lesions on my body that offered an easy way under my skin so that was something of a mystery. Its origin was mysterious in another way. Recent studies show that MRSA can originate in food animals and pass from animal handlers into the community. So the bacteria might have come from animals. But the bug could also have come from humans. Whether the antibiotics were used in medicine or food animals, overuse could have made my bug more resistant and virulent.
Stay Within the Lines
When I was finally released from the emergency room, I was given a prescription for an oral antibiotic and counseled to take every pill…which I, of course, planned to do. But as I walked out of the hospital into the warm summer night, I couldn’t help but worry that the antibiotic wouldn’t work.
“Draw a line around the red patch,” the doctor had said. “If it moves outside the lines, let me know.” If that happened, the doctors would have to turn to other drugs and by that time my red patch could have spread all over my torso and become very difficult to treat.
I was lucky. The hospital drip and the oral antibiotics did the trick. The red patch stayed within the lines, then faded, and I went on with my life.
There was nothing very dramatic about my experience. The antibiotics worked. We are so accustomed to our miracle drugs that it is hard to feel surprised or properly appreciative when they do what we expect. But my brush with (maybe) MRSA did leave an impression. I’ve gone back to working on antibiotic use in agriculture with a new sense of urgency. We need to curb antibiotic overuse in every setting, including feedlots, poultry houses, and swine barns.
No one knows where this infection came or when it might return. Next time, if there is one, I don’t want to be sitting in an emergency room as the red patch moves beyond the lines and can’t be stopped.